

PhageBiotix™ FOP is a revolutionary new proprietary formula containing lytic bacteriophages. It is intended for use as a dietary ingredient to help maintain healthy gut microflora and to increase resilience against pathogenic E. coli, Salmonella, and L. monocytogenes – bacteria associated with GI distress. The product contains a proprietary blend of naturally occurring lytic bacteriophages that, when taken regularly, encourage beneficial microbiome communities and help maintain resilient gut microflora. Published scientific research has shown PhageBiotix™ FOP to be very effective in eliminating major foodborne pathogens without disturbing the indigenous microbiota.
PhageBiotix™ FOP is the choice to help maintain healthy gut microflora because it:
Published scientific studies conducted by leading academic and industry researchers worldwide have demonstrated that PhageBiotix™ FOP is highly effective in eliminating its targeted foodborne pathogens while leaving the indigenous microbiota largely unaffected. Selected excerpts from these studies are provided below.
References:
1. Cieplak, T., et al. (2018). "A bacteriophage cocktail targeting Escherichia coli reduces E. coli in simulated gut conditions, while preserving a non-targeted
representative commensal normal microbiota." Gut Microbes: 01-19.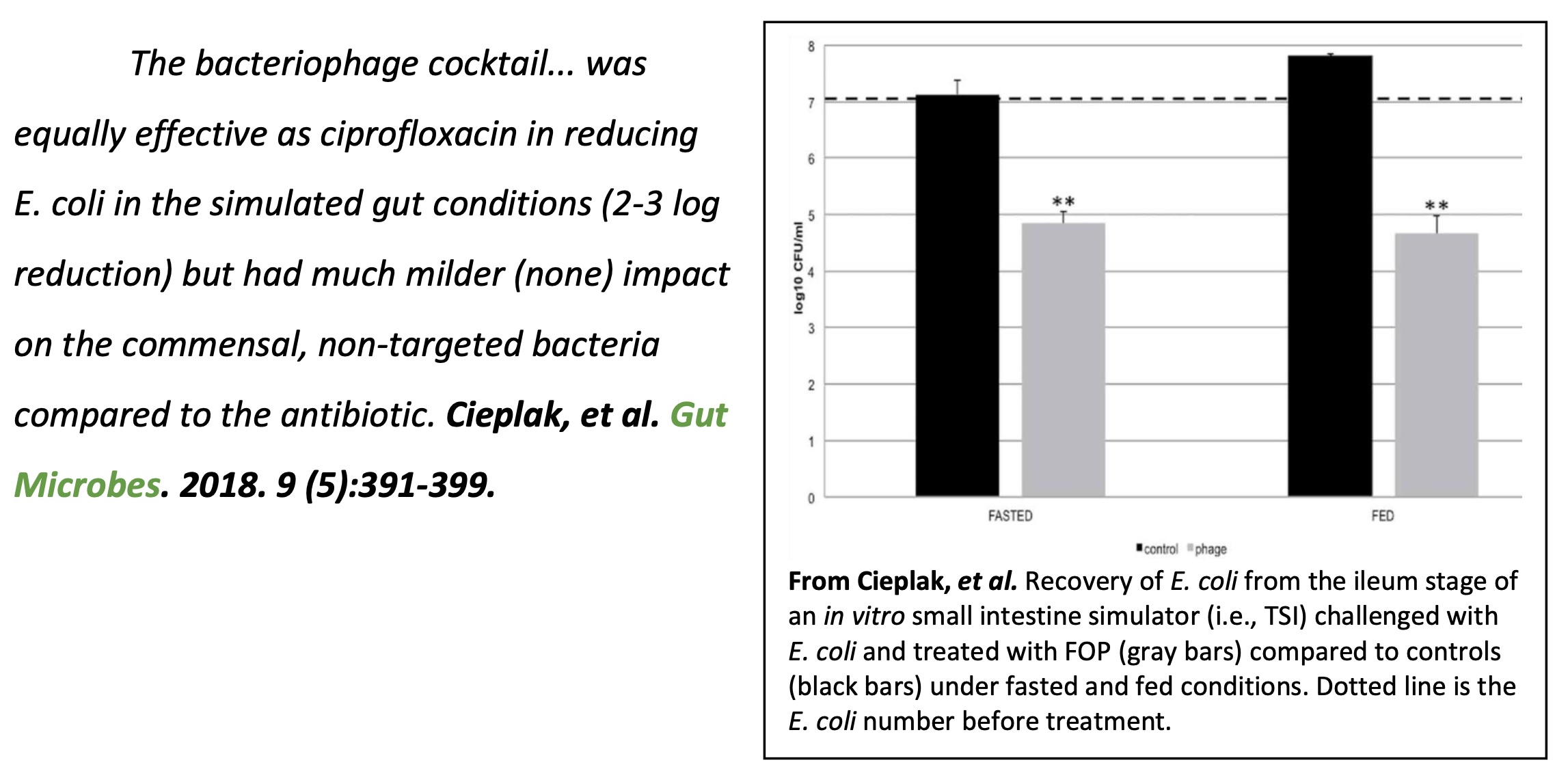
2. Moye, Z. D., et al. (2019). "A bacteriophage cocktail eliminates Salmonella
Typhimurium from the human colonic microbiome while preserving cytokine signaling and preventing attachment to and invasion of human cells by Salmonella in vitro." J Food Prot 82(8): 1336-1349.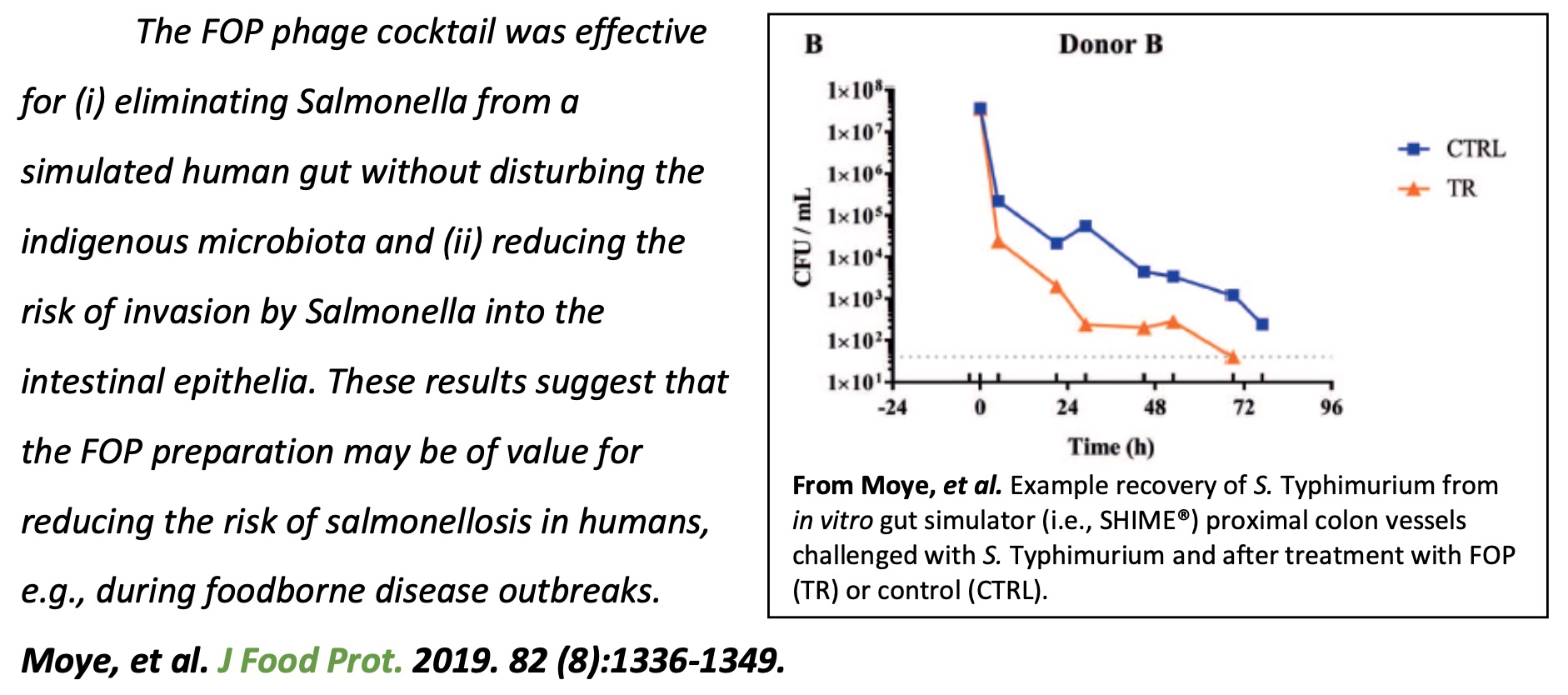
3. Dissanayake, U. et al. (2019). "Bacteriophages reduce pathogenic Escherichia coli counts in mice without distorting gut microbiota." Front Microbiol 10: 1984.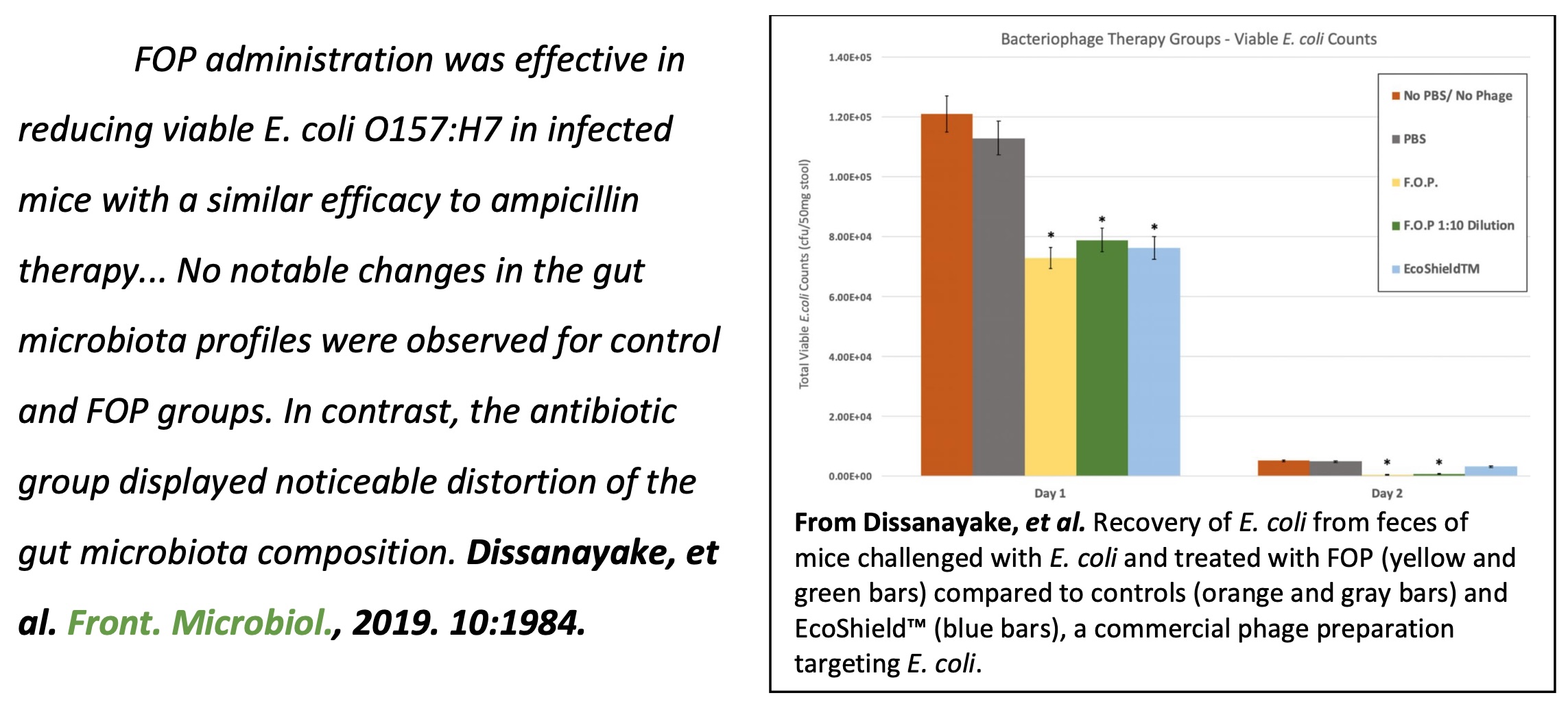
4. Jakobsen, R. R. et al. (2022). "A bacteriophage cocktail significantly reduces Listeria monocytogenes without deleterious impact on the commensal gut microbiota under simulated gastrointestinal conditions." Viruses 14(2).
PhageBiotix™ FOP is manufactured in SQF-certified cGMP (21 CFR 117 compliant) facility in the United States.
These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.





